
For example, a sample with a mass concentration of 100 mg/l will have a volume concentration of 38 µl/l. To convert from ppm by mass to ppm by volume, divide by the density of the particles. For example, a sample with a volume concentration of 25 µl/l will have a mass concentration of 25*2.65 = 66 mg/l.

For mineral grains (clay, silt and sand sizes), this will typically be 2.65 g/cm3. To convert from ppm by volume to ppm by mass, multiply by the density of the particles. However, if ppm is expressed as THE MASS of particles in a unit volume of water, then ppm BY MASS is equal to mg/l. If ppm is expressed as THE VOLUME of particles to a unit volume of water, then ppm BY VOLUME is equal to µl/l. But what parts? The amount of particles in a suspension can be expressed as the total volume OR total mass of particles in a unit volume of water AND THESE TWO NUMBERS WILL ONLY BE THE SAME IF THE DENSITY OF THE PARTICLES IS 1 g/cm3. Therefore, we need to dilute 0.005 L (or 5 mL) of stock solution to a final volume of 1 L to get a 200 ppm NaOH solution.Ppm means parts-per-million. We can fill in the numbers for all the variables except for the volume of stock solution:īy rearranging the equation, we will find the volume of stock solution required:
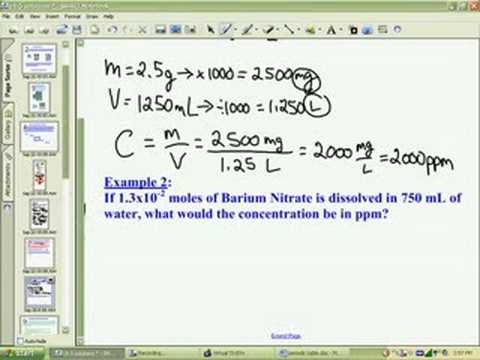

The "part" can be any unit, but when mixing solutions, ppm will usually represent weight units. So, what is ppm? And how can something called "parts per million" be represented by mg/L? Parts per million indicates the number of "parts" of something in a million "parts" of something else. ppm of pollutants in air (which uses μL of pollutant per L of air).ppm of a nutrient in soil (which uses mg nutrient per kg of soil) and.You will need a different ppm calculation if you are using it in other contexts, such as: Mixing fertilizer solutions for horticulture and.Maintenance of chemical levels in aquariums.Measurements of drinking water quality.Some situations where you may need to convert ppm to molarity include: There are 6.0221409 * 10 23 molecules or particles in one mole. Many concentration calculations use the mole units because we're dealing with a great number of molecules. On the other hand, molarity is molar concentration, meaning that it tells you how many moles of molecules are in one L of water. Did you know that ppm is used in different ways depending on the context? When dealing with dilute solutions, 1 ppm can be approximated as 1 mg of substance per liter of water, or 1 mg/L. Both ppm (parts per million) and molarity are measures of concentration.
